

Normec Hybeta ist Ihr Spezialist für mikrobiologische und technische Hygiene im Gesundheitswesen. Wir prüfen unabhängig, beraten praxisorientiert und sorgen so für die Sicherheit Ihrer Patienten. Die hygienischen Anforderungen steigen kontinuierlich, entsprechend wachsen auch die Herausforderungen in medizinischen Einrichtungen.
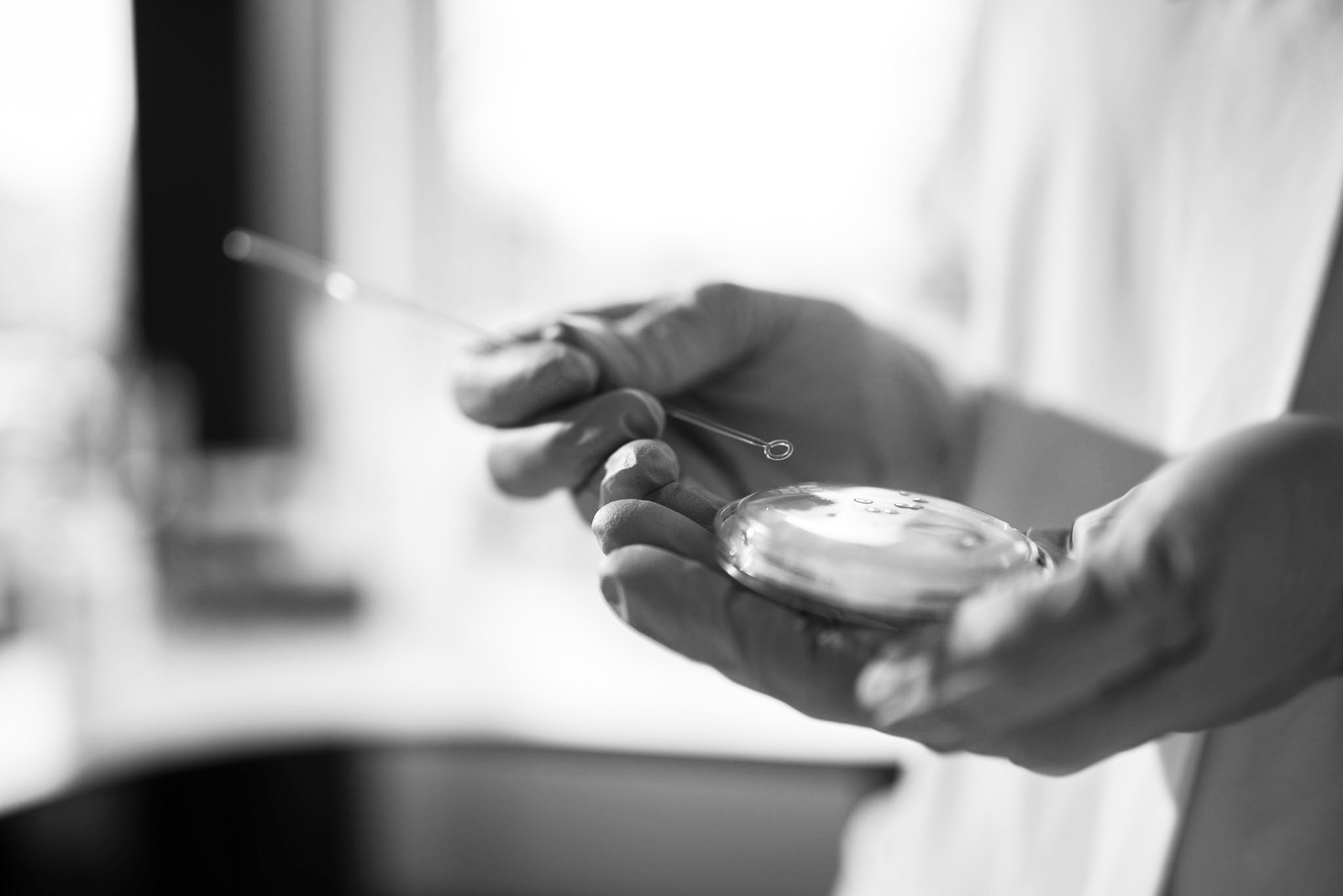
Unabhängige Prüfungen durch ein unabhängiges Prüflabor.
Wir sind nach DIN EN ISO/IEC 17025 als unabhängiges Prüflabor akkreditiert.
Wir liefern unseren Kunden behördenkonforme Berichte.


Vom 12.-15.Mai 2024 findet der 17. Kongress für Krankenhaushygiene in Bonn statt.

Heute vor zwei Wochen war es so weit – in diesem Jahr hat uns der Februar einen Tag mehr geschenkt.

Die European Medicines Agency (EMA) hat aufgrund der COVID-19-Pandemie ein Update zu der GMP-Compliance veröffentlicht.
Auf unserem Infoblatt.
Wenn Sie gerne kostenlos die schnelle und papierlose Übermittlung von Befunden nutzen möchten, dann senden Sie uns das entsprechende Anforderungsformular mit Ihrer Beauftragung zu.
Wir bieten Ihnen die Möglichkeit, regelmäßig wiederkehrende Untersuchungen in sog. Intervallen zu hinterlegen, damit wir Sie zum passenden Zeitpunkt per E-Mail an die anstehende Prüfung erinnern können. Beauftragen Sie uns, wenn Sie diese Intervall-Buchung wünschen, damit Ihre Arbeit schnell und zuverlässig erledigt werden kann.